There are quite a few articles and great resources for developing and conducting a comprehensive Gap Analysis around the compliance details defined in the United States Pharmacopeia (USP) chapter . Despite the title of this article we do not intend on simply repeating those articles, instead we want to address details for performing a gap analysis focused on the business side of the compliance equation. USP 800 is going to financially impact your pharmacy’s future business model so this article will focus on five important details that define the “why” for considering the capital investment. In this article we will offer up some examples that show how the gap analysis is a simple and effective analytical process that can help you understand if the capital investment is advantageous. It is difficult to design a “one size fits all” hazardous drug handling compliant facility without taking into consideration your pharmacy's specific business model, but towards the end of this article we will propose a financial model based on some generic assumptions to put capital investment into business perspective.
View the Full Guide for USP 800 Gap Analysis
USP 800 Gap Analysis
Related Resources

Video
Telescoping Base Stand Assembly for Your Class II Biosafety Cabinet
This video shows how to assembly a telescoping base stand and safely install your Class II Biosafety Cabinet on the stand.

White Paper
Surface Decontamination in a Biosafety Cabinet
Learn the right techniques for surface decontamination in a biosafety cabinet. Protect personnel, products, and environments. Download NuAire’s expert white paper today.

Article
Smoke Tests NSF/ANSI 49
From NSF International, watch this video to learn how accredited certifiers perform a four different times of smoke tests based on NSF/ANSI 49: Biosafety Cabinetry – Design, Construction, Performance, and Field Certification. Source – NSF International

Video
Therapeutics to Combat Aggressive Cancer
Dr. Aleksandra Dukic, postdoctoral researcher, Institute for Cancer Research, Oslo University Hospital, describes a novel target for inhibiting glioblastoma cancer invasion into surrounding brain tissue and the importance of this work in addressing the need for innovative therapeutics to combat this aggressive cancer. Hear how the culture and analysis of glioblastoma patient-derived cells are vital […]
Video
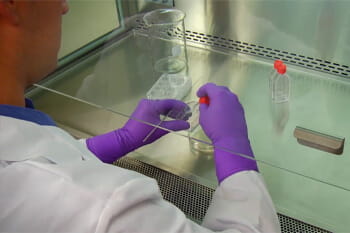
Understanding the Biosafety Cabinet
Biosafety Cabinets (BSCs) a are designed to remove or minimize exposures to hazardous biological materials and particulates.

Video
Telescoping Base Stand Assembly
Learn how to assemble telescoping base stands with simple tools. Follow step-by-step instructions for secure assembly and mounting of your biosafety cabinet.
Video
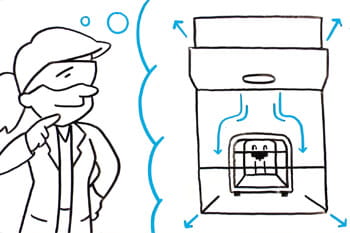
Tailor Made Biosafety Cabinets for Automation
Gain insight into the custom biosafety cabinet process, and collaborate with engineers to design tailor-made solutions for specific lab workflows, ensuring the safe containment of automated equipment while optimizing performance.

White Paper
Tips for Purchasing a Biosafety Cabinet
The white paper is a guide for principal investigators when purchasing a biosafety cabinet.

Article
Setting Up the Lab
Setting up a lab requires strategic design for safety and efficiency, and the choice of appropriate biosafety cabinets based on the risk level of materials handled.

Webinar
USP Compliance USP 795 Facility Requirements
Bryan Prince reviews the timeline for USP 795 compliance, QC processes for nonsterile compounding, staff training steps, and more.

White Paper
Standard Operating Procedures (SOPs) for Biosafety Cabinet Use
Learn why and how to create standard operating procedures (SOPs) for biosafety cabinet use to improve lab safety, compliance, and workflow consistency. Download the full guide today.

Ebook
10 Steps to Cleaning a Spill in a Biosafety Cabinet
Discover how to safely handle spills in a Biosafety Cabinet with NuAire's eBook, "Ten Easy Steps for Cleaning a Spill in the Biosafety Cabinet." This guide provides clear instructions on spill management, decontamination, and adherence to laboratory safety standards.
Related Products

LabGardu00ae ES NU-540 Class II, Type A2 Biosafety Cabinet

LabGardu00ae ES AIR NU-543 Class II, Type A2 Biosafety Cabinet

LabGardu00ae ES NU-427 Class II, Type B1 Biosafety Cabinet

LabGardu00ae ES AIR Limited NU-545 Class II, Type A2 Biosafety Cabinet

LabGard TE (Total Exhaust) NU-560 Class II, Type B2 Biosafety Cabinet

LabGard CYTO NU-581 Class II, Type A2 Biosafety Cabinet

LabGardu00ae ES FH NU-565 Class II, Type B2 Biosafety Fume Hood

LabGardu00ae ES NU-677 Class II, Type A2 Animal Handling Biosafety Cabinet

LabGardu00ae ES NU-610 Dual Access Class II, Type A2 Animal Handling Biosafety Cabinet

LabGard LP NU-640 Class II, Type A2 Animal Handling Biosafety Cabinet

LabGardu00ae ES NU-813 Class I Containment Ventilated Enclosure

AutoLabGard NU-L125 Mini-Room Class II, Type A2 Biosafety Cabinet

AireGard ES (Energy Saver) NU-140 Vertical Laminar Flow Hood

AireGard ES (Energy Saver) NU-340 Console Horizontal Laminar Flow Hood

AireGard ES (Energy Saver) NU-240 Bench Top Horizontal Laminar Flow Hood

PharmaGard NU-NTE800 Total Exhaust Compounding Aseptic Containment Isolator

PharmaGard ES NU-PR797 Compounding Aseptic Isolator



