
USP Compliance Staff Training for USP

Fred Massoomi, PharmD, FASHP, reviews staffing considerations under USP <800>, including topics such as the value of didactic and experiential training and how to establish training by position. Original content from Pharmacy Purchasing & Products – www.pppmag.com
USP Compliance Staff Training for USP
Related Resources

Webinar
USP Compliance USP 795 Facility Requirements
Bryan Prince reviews the timeline for USP 795 compliance, QC processes for nonsterile compounding, staff training steps, and more.
Webinar
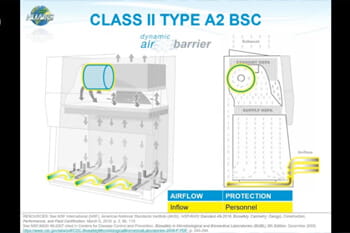
Working With Not Against Your Biosafety Cabinet
Learn how to optimize the use of your biosafety cabinet with expert tips on setup, maintenance, and operation to ensure maximum safety and efficiency.

White Paper
Understanding Compounding Isolators
This white paper discusses restricted access barrier systems (RABS) in the pharmacy, how they operate, cleaning considerations, maintenance, and more.

White Paper
USP 800 Gap Analysis
USP 800 is going to financially impact your pharmacy’s future business model so this white paper focuses on five important details that define the "why" for considering a capital investment.

Webinar
Cleanroom Layout Design Under USP 800
Learn the necessary elements to establish a cleanroom layout under USP 800.

Article
Use of Pharmacy Compounding Isolator Onboard US Navy Ship
NuAire pharmacy compounding isolators are used onboard ships of the United States Navy, particularly the hospital ships USNS Mercy and USNS Comfort.

Webinar
USP Compliance Preparing for Cleanroom Certification
Jim Wagner addresses how to work with a certifier, typical issues that arise during the certification process, how to respond to the certification report, and what follow-up measures should be taken.

Webinar
USP Compliance PEC Cleaning and Decontamination
Fred Massoomi outlines how to establish standard operating procedures (SOPs) for cleaning PECs, the level of detail required for these SOPs, and staff training requirements.

Infographic
USP 797 and USP 800 PEC Requirements
Follow this flowchart for an easy method to identify what Primary Engineering Control (PEC) or Containment Primary Engineering Control (C-PEC) your pharmacy needs.

General Technical Bulletin
Using a Chemo Pad in a Class II, Type A2 Biosafety Cabinet
The bulleting entitled Chemo Pad Containment Capability of a Biosafety Cabinet offers insight into the performance capabilities of a biosafety cabinet (BSC) using a chemotherapy pad on the work surface.

Video
Testing Your Adjustable Base Stand
Learn how to test your adjustable base stand on your PharmaGard Compouding Isolator
Webinar
USP Compliance Safe Implementation of PECs Under USP 797 and 800
Jim Wagner reviews the impact of the changes to USP <797> with regard to PEC implementation & USP <800> requirements for containment.
